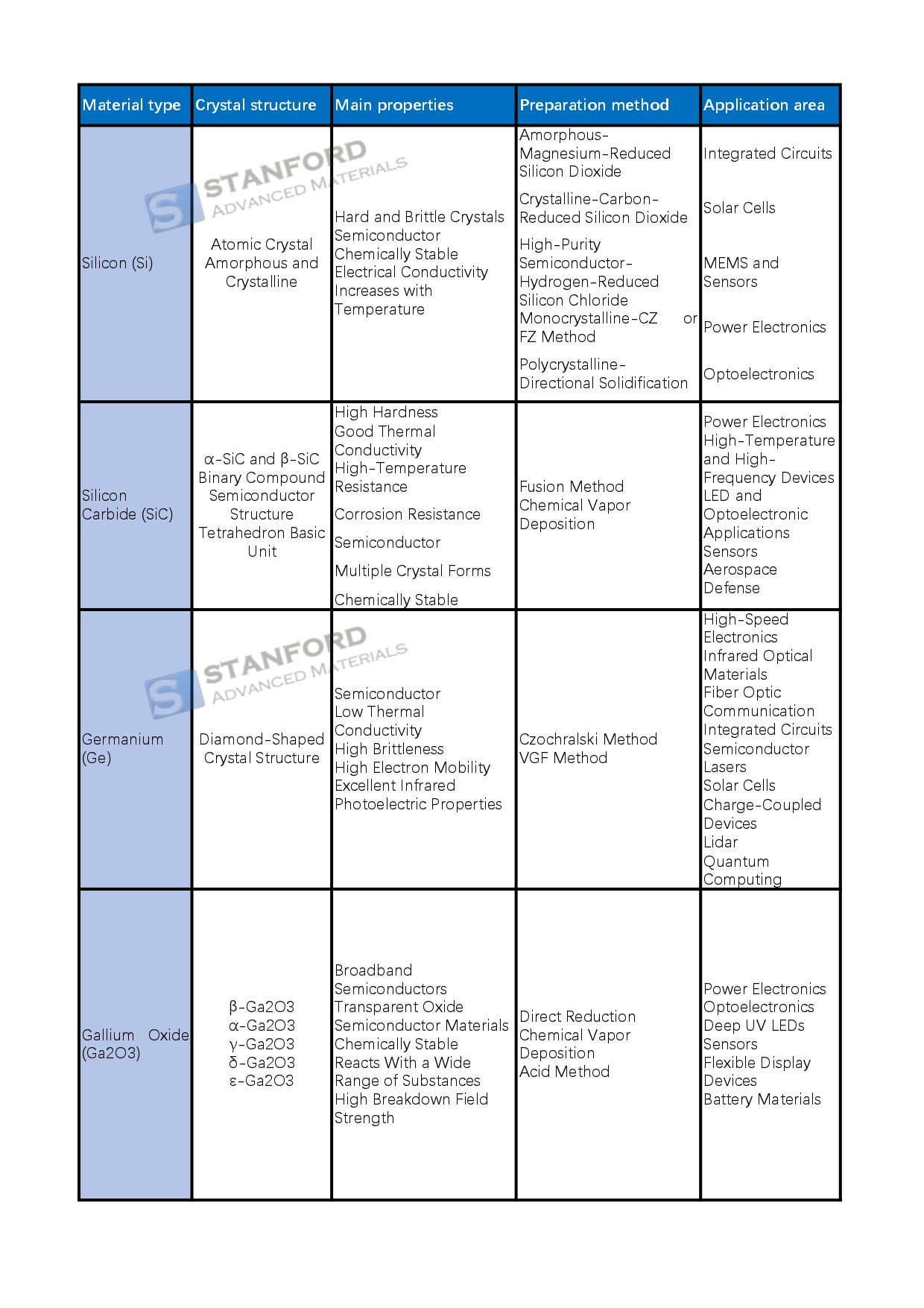
The Essential List Of Electronic Materials
1 Introduction
Electronic materials play a vital role in modern electronics. They function as fundamental components that determine the performance of a wide range of devices, from smartphones and laptops to aerospace and medical instruments. These materials dictate the electrical, thermal, mechanical and optical properties and, consequently, set the operational limits of the devices.
The development and innovation of electronic materials have driven steady progress in the electronics industry. Given the increased demand for smaller, faster, energy‐efficient and highly reliable products, the search for advanced materials has become urgent. The sections below examine silicon, silicon carbide, germanium, gallium compounds, carbon‐based materials, conductive and insulative materials and magnetic materials. By reviewing their quantifiable properties, specific manufacturing methods and applications, a clear view of their importance and potential for shaping future electronics is obtained.
2 Silicon and Related Materials
Silicon (Si) has atomic number 14. It is a hard, brittle crystalline material with quasi-metallic and semiconducting properties. Silicon occurs in both amorphous and crystalline forms and is abundant in the Earth’s crust. It has four valence electrons and shows limited chemical reactivity at room temperature. Its electrical conductivity increases with temperature and it is used in various electronic devices.
Amorphous silicon is typically produced by reducing silicon dioxide with magnesium. Crystalline silicon is fabricated in an electric furnace by reducing silicon dioxide with carbon. High‐purity semiconductor silicon is achieved by hydrogen reduction of silicon chloride. Monocrystalline silicon is mainly grown using the Czochralski process or by zone melting, whereas polycrystalline silicon is produced through directional solidification.
Silicon is used extensively in the electronics industry. It serves as a chip substrate in integrated circuits and is ideal for transistor production. Monocrystalline, polycrystalline and thin silicon films are employed in solar cells. In micro‐electromechanical systems (MEMS) and sensors, silicon is used for accelerometers, gyroscopes and pressure sensors. In power electronics, it is applied to manufacture power semiconductors such as MOSFETs and IGBTs. In optoelectronics, silicon photodiodes are utilised in optical communication systems. Its benefits include abundant reserves, controlled purity and low cost; however, its performance declines at high temperatures and its narrow bandgap restricts its use in high‐speed, high‐frequency applications.
Read more on this: Essential Electronic Materials: Part 1 – Silicon
Fig.1 Silicon Products
3 Silicon Carbide (SiC)
Silicon carbide (SiC) is a wide bandgap semiconductor known for its high hardness, good thermal conductivity and resistance to high temperatures and corrosion. It exists in several crystal forms, predominantly α-SiC and β-SiC. Industrially produced SiC may show varied colours due to the presence of impurities. Doping methods are used to adjust its energy levels and physical properties.
The primary production methods for silicon carbide include melting and chemical vapour deposition (CVD). In the melting process, silicon carbide is formed by mixing silicon and graphite at high temperatures followed by controlled cooling. In the CVD process, SiC is deposited from a gas-phase reaction on a substrate, typically to produce high-quality thin films.
SiC is applied in numerous fields. In power electronics it is used in power conversion devices that improve the efficiency of electric vehicles and renewable energy systems. In high-temperature and high-frequency devices, it is used in 5G base stations and military electronics, given its capability to operate under extreme conditions. It is also employed in LED and optoelectronic applications and in sensors that monitor high temperatures, pressures and radiation. Additionally, its performance makes it suitable for aerospace and defence applications.
The advantages of silicon carbide include excellent performance under high temperature, pressure and frequency conditions, which leads to high energy conversion efficiency. However, higher costs and a complex manufacturing process remain challenges, and ongoing efforts aim to improve device reliability.
Read more on this: Essential Electronic Materials: Part 2 – Silicon Carbide
Fig. 2 Silicon Carbide Chip
4 Germanium and Related Materials
Germanium (Ge) is a semiconductor element with atomic number 32 and a diamond-like crystal structure. It has a density of 5.323 g/cm³, a melting point of 937.4°C and a boiling point of 2833°C. As a semiconductor, germanium has a bandgap of 0.66 eV and demonstrates good electrical conductivity at low temperatures. Its optoelectronic properties are particularly useful in the infrared range, although its low thermal conductivity and brittleness limit some applications.
Germanium is generally produced as a single crystal using the Czochralski process or the vertical gradient freeze (VGF) method. In the Czochralski procedure, molten germanium is drawn out to form a single crystal. The VGF method regulates the temperature gradient to maintain crystal quality and purity.
Germanium is used in high-speed electronics. It finds application in high-frequency transistors and wireless communications due to its high electron mobility, which supports rapid signal processing. It is an important optical material in the infrared range and is utilised in infrared detectors and imaging systems. It is also doped into optical fibres to improve performance. In integrated circuits, germanium is combined with silicon to form alloys that further enhance performance. Additionally, germanium is used in semiconductor lasers, charge-coupled devices (CCDs) and LIDAR, and it holds potential in quantum information technologies.
Germanium’s benefits include high electron mobility, a low bandgap, high infrared transparency and good compatibility with silicon. Its disadvantages are low thermal conductivity, high cost, difficulties in crystal growth and a limited industrial production scale.
Read more on this: Essential Electronic Materials: Part 3 – Germanium
Fig. 3 Germanium for Chip Production
5 Gallium Compounds
Gallium oxide (Ga2O3) is a wide bandgap semiconductor that exists in several crystal forms such as β-Ga2O3 and α-Ga2O3. It is chemically stable and reacts with various substances. It is produced by processes including direct reduction, gas-phase deposition and acid-assisted methods. It is used in power electronics, optoelectronic devices (for example, ultraviolet detectors and deep ultraviolet LEDs), sensors and other applications.
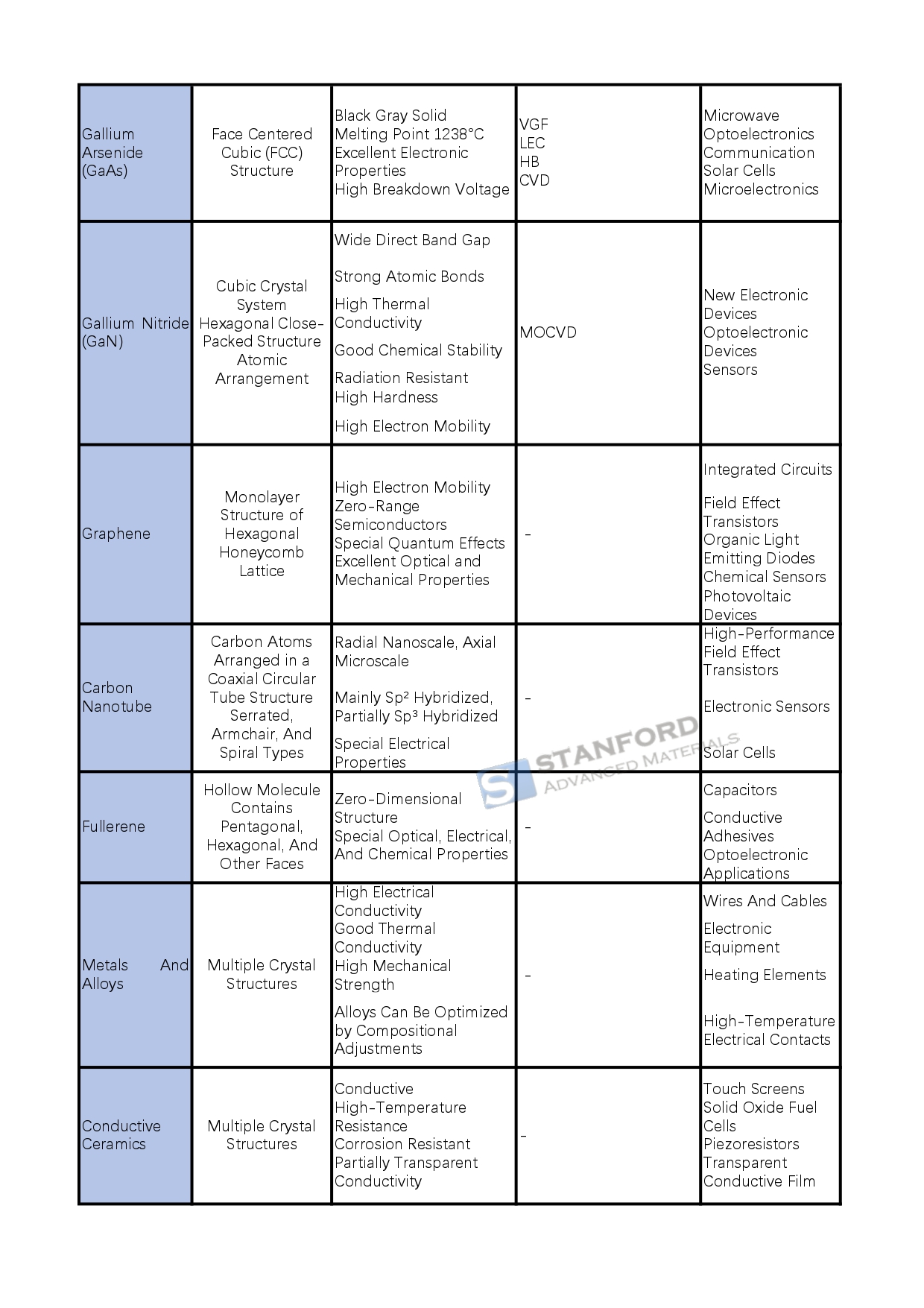
Gallium arsenide (GaAs) is a black-grey solid with a melting point of 1238 °C. It is a significant III–V compound semiconductor with a cubic crystal structure. It offers excellent electronic performance, low noise and high breakdown voltage. It is manufactured by methods such as Vertical Gradient Solidification (VGF), Liquid Encapsulated Czochralski (LEC), Horizontal Bridgman (HB) and Chemical Vapour Deposition (CVD). It is used in microwave systems, optoelectronics, communications, solar cells, microelectronics and related fields.
Gallium nitride (GaN) consists of a crystal lattice of gallium and nitrogen atoms. It has a wide direct bandgap, high hardness, high thermal conductivity and good chemical stability. It is produced by metal organic chemical vapour deposition (MOCVD) along with other methods. GaN is employed in high-performance high-frequency devices, optoelectronic devices such as short-wavelength LEDs and sensors, thereby supporting technological progress.
Read more on this: Essential Electronic Materials: Part 4 – Gallium Compounds
Fig. 4 Gallium Oxide Semiconductor Materials
6 Carbon-based Materials
Graphene is a single-atom-thick structure where carbon atoms are sp²-hybridised to form a hexagonal lattice. It exhibits excellent optical, electronic and mechanical properties. Its high electron mobility makes it applicable in integrated circuits, field-effect transistors, organic LEDs, chemical sensors and optoelectronic devices.
Carbon nanotubes (CNTs) are cylindrical structures composed of carbon atoms. They show distinct electrical properties and are classified as either metallic or semiconducting. They are employed in high-performance field-effect transistors, electronic sensors and solar cells, thereby enhancing device performance.
Fullerenes (Fullerenes) are hollow molecules consisting of carbon atoms, such as C60. They possess specific optical, electronic and chemical properties. They are used in capacitors, conductive adhesives and optoelectronic applications, and they can improve electrode conductivity and energy storage density.
Read more on this: Essential Electronic Materials: Part 5 – Carbon-based Materials
Fig. 5 Graphene
7 Conductive and Insulative Materials
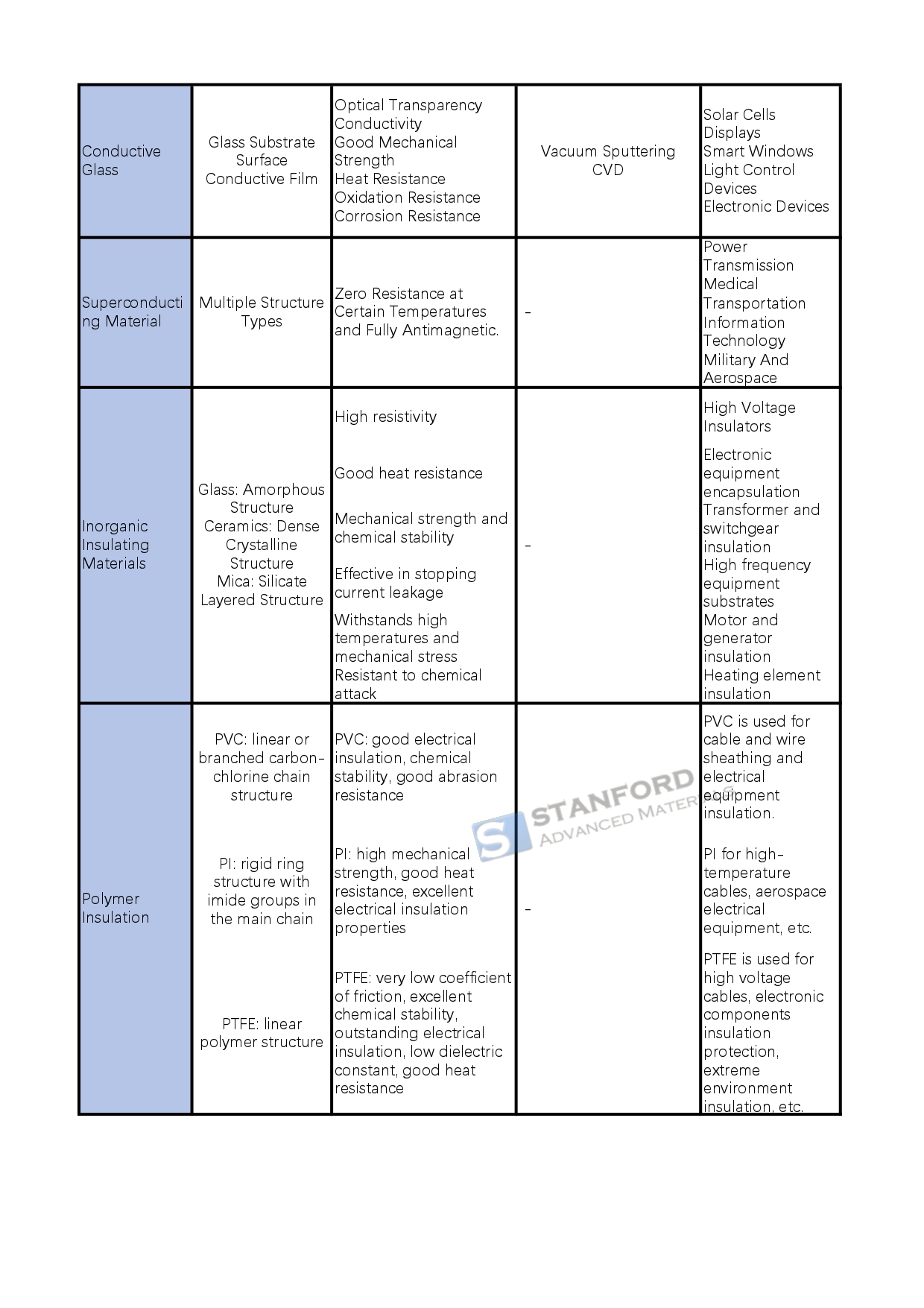
Conductive materials include metals and alloys that easily generate free electrons to conduct electricity. They show high electrical conductivity, good thermal conductivity and satisfactory mechanical strength. For example, copper, silver and aluminium alloys are widely used in wires, cables and electronic devices. Conductive ceramics pass electricity via ion migration or electron transport and offer high temperature and corrosion resistance, as seen with indium tin oxide (ITO) in touchscreens. Conductive glass, which comprises a glass substrate with a conductive surface layer, is used in solar cells and displays. Superconducting materials exhibit zero resistance at certain temperatures and complete diamagnetism, and they are applied in power transmission, medical instruments, transport and information technology.
Insulative materials comprise inorganic substances such as glass, ceramics and mica. These materials have a high specific resistance, good heat resistance, solid mechanical strength and chemical stability and are used for electrical insulation. For instance, glass is used in high-voltage insulators and ceramics in transformer insulation. Polymeric insulators such as PVC, polyimide (PI) and polytetrafluoroethylene (PTFE) are selected for various electronic applications and environments.
Read more on this: Essential Electronic Materials: Part 6 – Conductive and Insulative Materials
Fig. 6 Ceramic Electronic Materials
8 Magnetic Materials
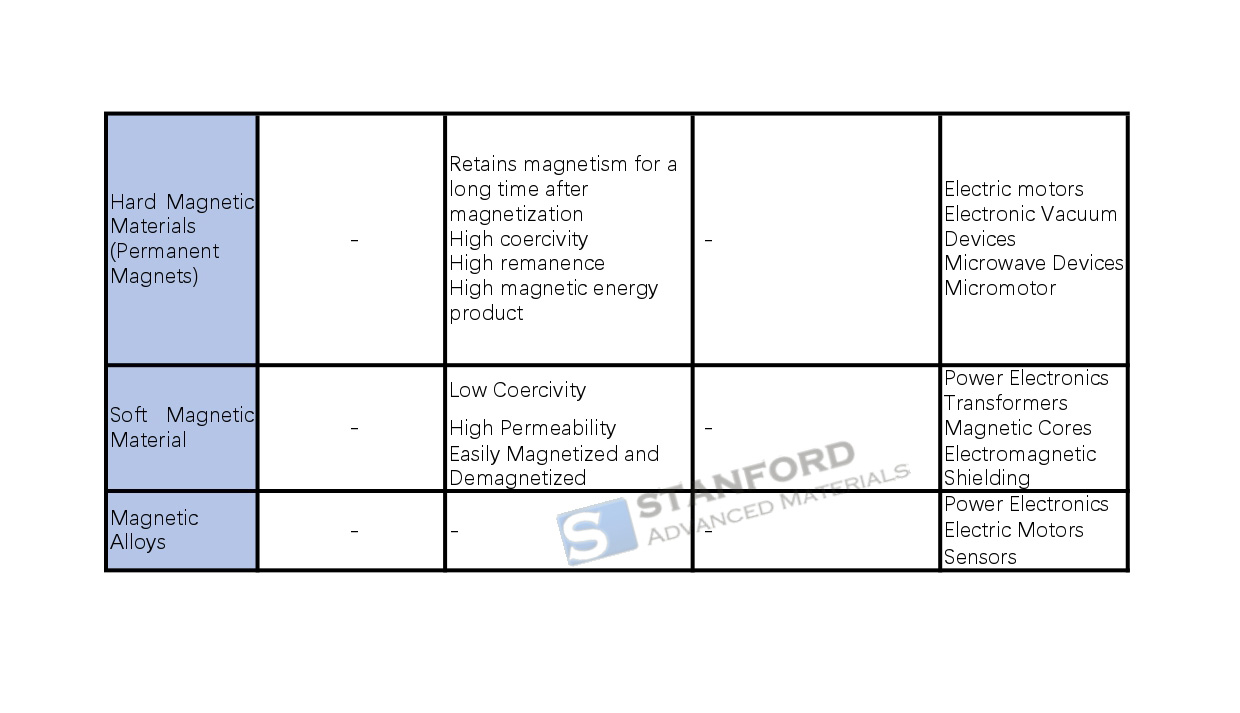
Hard magnetic materials, also known as permanent magnets, include Alnico alloys, titanium-cobalt alloys and rare-earth cobalt magnets. They retain magnetism for long periods after magnetisation and display high coercivity, remanence and magnetic energy product. These materials are used in electric motors, vacuum electronic devices and microwave equipment.
Soft magnetic materials, such as silicon steel and ferrite, exhibit low coercivity and high permeability. They can be magnetised and demagnetised easily. They are common in transformers, motors, chokes and electromagnetic shielding, thereby reducing energy losses and increasing efficiency.
Magnetic alloys, composed of iron and other metals, are divided into soft and hard types. They are used in electronics, communications and energy applications. For example, neodymium-iron-boron (NdFeB) is utilised in electric motors, and aluminium-nickel-cobalt alloys find use in high-temperature environments. The magnetic properties — including permeability, saturation flux density and coercivity — are critical for the performance of power supplies, transformers, data storage devices, motors, sensors and wireless communication systems.
Read more on this: Essential Electronic Materials: Part 7 – Magnetic Materials
Fig. 7 NdFeB Magnets
9 Conclusion
In conclusion, electronic materials form the foundation of modern electronics. Although silicon has predominated historically, materials such as silicon carbide and gallium compounds are gaining importance. Carbon-based materials offer additional possibilities. Conductive, insulative and magnetic materials are also essential in the construction of devices. Research efforts are focused on improving performance and reliability. Continuous research and material optimisation meet existing requirements and enable new technological applications. The systematic study of these materials is essential for progress in the electronics industry.
Stanford Advanced Materials (SAM) is a key supplier of high-quality electronic materials that supports critical applications with reliable material solutions.

 Bars
Bars
 Beads & Spheres
Beads & Spheres
 Bolts & Nuts
Bolts & Nuts
 Crucibles
Crucibles
 Discs
Discs
 Fibers & Fabrics
Fibers & Fabrics
 Films
Films
 Flake
Flake
 Foams
Foams
 Foil
Foil
 Granules
Granules
 Honeycombs
Honeycombs
 Ink
Ink
 Laminate
Laminate
 Lumps
Lumps
 Meshes
Meshes
 Metallised Film
Metallised Film
 Plate
Plate
 Powders
Powders
 Rod
Rod
 Sheets
Sheets
 Single Crystals
Single Crystals
 Sputtering Target
Sputtering Target
 Tubes
Tubes
 Washer
Washer
 Wires
Wires
 Converters & Calculators
Converters & Calculators
 Write for Us
Write for Us









 Chin Trento
Chin Trento



